Crystals Can Have _____ Bonds Between Their Atoms.
Metals account for most ii thirds of all the elements and most 24% of the mass of the planet. They are all effectually us in such forms every bit steel structures, copper wires, aluminum foil, and gold jewelry. Metals are widely used because of their backdrop: strength, ductility, high melting betoken, thermal and electrical electrical conductivity, and toughness.
These backdrop too offer clues as to the structure of metals. Equally with all elements, metals are composed of atoms. The force of metals suggests that these atoms are held together by strong bonds. These bonds must likewise allow atoms to move; otherwise how could metals be hammered into sheets or drawn into wires? A reasonable model would exist one in which atoms are held together past strong, but delocalized, bonds.
Bonding
Such bonds could be formed between metal atoms that have low electronegativities and do not concenter their valence electrons strongly. This would permit the outermost electrons to exist shared past all the surrounding atoms, resulting in positive ions (cations) surrounded by a sea of electrons (sometimes referred to as an electron cloud).
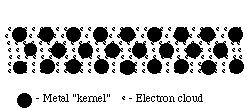
Because these valence electrons are shared by all the atoms, they are non considered to exist associated with any 1 atom. This is very different from ionic or covalent bonds, where electrons are held by one or two atoms. The metal bond is therefore strong and uniform. Since electrons are attracted to many atoms, they have considerable mobility that allows for the good estrus and conductivity seen in metals.
Above their melting signal, metals are liquids, and their atoms are randomly arranged and relatively free to move. However, when cooled below their melting point, metals rearrange to form ordered, crystalline structures.
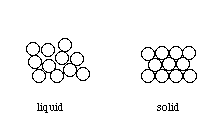
Crystals
To form the strongest metallic bonds, metals are packed together as closely every bit possible. Several packing arrangements are possible. Instead of atoms, imagine marbles that demand to be packed in a box. The marbles would be placed on the bottom of the box in neat orderly rows then a second layer begun. The second layer of marbles cannot be placed directly on top of the other marbles and then the rows of marbles in this layer move into the spaces betwixt marbles in the showtime layer. The showtime layer of marbles tin can be designated as A and the second layer as B giving the two layers a designation of AB.
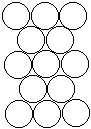 | 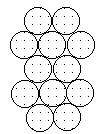 | 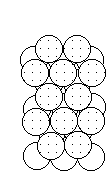 |
|---|---|---|
| Layer "A" | Layer "B" | AB packing |
Packing marbles in the tertiary layer requires a decision. Once again rows of atoms will nest in the hollows betwixt atoms in the second layer simply ii possibilities exist. If the rows of marbles are packed so they are directly over the offset layer (A) so the arrangement could be described equally ABA. Such a packing system with alternate layers would exist designated every bit ABABAB. This ABAB organisation is called hexagonal shut packing (HCP).
If the rows of atoms are packed in this third layer so that they exercise not prevarication over atoms in either the A or B layer, so the 3rd layer is chosen C. This packing sequence would be designated ABCABC, and is also known as face-centered cubic (FCC). Both arrangements give the closest possible packing of spheres leaving simply near a quaternary of the available space empty.
The smallest repeating assortment of atoms in a crystal is called a unit cell. A third mutual packing arrangement in metals, the torso-centered cubic (BCC) unit cell has atoms at each of the 8 corners of a cube plus ane atom in the middle of the cube. Because each of the corner atoms is the corner of another cube, the corner atoms in each unit cell volition exist shared among eight unit of measurement cells. The BCC unit cell consists of a net total of two atoms, the ane in the center and viii eighths from the corners.
In the FCC arrangement, over again there are eight atoms at corners of the unit prison cell and ane atom centered in each of the faces. The atom in the face is shared with the side by side jail cell. FCC unit cells consist of 4 atoms, eight eighths at the corners and six halves in the faces. Table i shows the stable room temperature crystal structures for several elemental metals.
| Aluminum | FCC | | Nickel | FCC |
|---|---|---|---|---|
| Cadmium | HCP | | Niobium | BCC |
| Chromium | BCC | | Platinum | FCC |
| Cobalt | HCP | | Silver | FCC |
| Copper | FCC | | Titanium | HCP |
| Aureate | FCC | | Vanadium | BCC |
| Atomic number 26 | BCC | | Zinc | HCP |
| Lead | FCC | | Zirconium | HCP |
| Magnesium | HCP |
Unit prison cell structures determine some of the backdrop of metals. For example, FCC structures are more than probable to be ductile than BCC, (body centered cubic) or HCP (hexagonal close packed). Figure 4 shows the FCC and BCC unit cells. (Encounter Crystal Construction Activity)
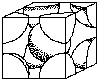 | 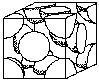 |
|---|---|
| Torso Centered Cubic | Face Centered Cubic |
As atoms of melted metallic brainstorm to pack together to form a crystal lattice at the freezing indicate, groups of these atoms form tiny crystals. These tiny crystals increase in size past the progressive addition of atoms. The resulting solid is not one crystal just actually many smaller crystals, chosen grains. These grains grow until they impinge upon adjacent growing crystals. The interface formed betwixt them is chosen a grain purlieus. Grains are sometimes large plenty to be visible under an ordinary light microscope or fifty-fifty to the unaided center. The spangles that are seen on newly galvanized metals are grains. (Run into A Particle Model of Metals Activity) Figure five shows a typical view of a metal surface with many grains, or crystals.

Crystal Defects:
Metallic crystals are non perfect. Sometimes there are empty spaces called vacancies, where an cantlet is missing. Another mutual defect in metals are dislocations, which are lines of lacking bonding. Figure 6 shows ane blazon of dislocation.
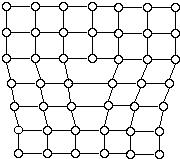
These and other imperfections, as well as the beingness of grains and grain boundaries, determine many of the mechanical properties of metals. When a stress is applied to a metallic, dislocations are generated and motion, allowing the metal to deform.
Dorsum to elevation
Source: https://depts.washington.edu/matseed/mse_resources/Webpage/Metals/metalstructure.htm
0 Response to "Crystals Can Have _____ Bonds Between Their Atoms."
Postar um comentário